Pfizer Set to Launch COVID 19 Vaccine for 5-year-old Kids
-
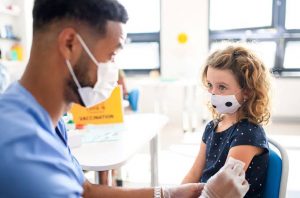
Pfizer announced on Monday that its COVID 19 vaccine works for children between the ages of 5 and 11
-
The company is currently seeking government authorization for the vaccine to be used on children within the age group
-
The vaccine made in collaboration with BioNTech is already available for anyone above the age of 12
Pfizer revealed on Monday that its COVID 19 vaccine has been confirmed effective and safe for children between the ages of 5 and 12.
Pfizer chairman and CEO Albert Bourla announced that the company is seeking approval from the US government for the vaccine to be used in children of those ages amid a surge in delta variant.
“We are eager to extend the protection afforded by the vaccine to this younger population, subject to regulatory authorization, especially as we track the spread of the delta variant and the substantial threat it poses to children,”
The Pfizer chairman stressed the need for vaccination following a 240 percent in pediatric cases of COVID 19 in the US.
2,268 participants ages between the ages of 5 and 12 took part in the company’s vaccine trial. Although the children also received two shots of the vaccine, a lesser dose was administered.
According to a report from the company, the vaccine had no major side effects and neutralized antibody responses.
Results from two other ongoing trials are ongoing (ages 2 to 5 and 6 months to 2) are currently being carried out and are expected before year’s end.
Read also: Nigeria Bans Brazil and South Africa, Removes India Restriction
The vaccine which was developed in collaboration with BioNTech will also seek approval for those age groups if the trials come out successful.
Follow Naija.fm on our social media handles Facebook, Instagram, and Twitter to keep up with trending news, breaking news, entertainment news.