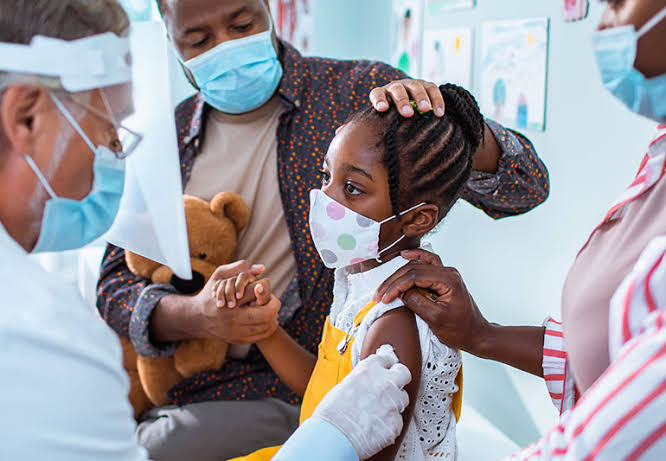
BioNTech Set to Launch COVID 19 Vaccine for Children
-
BioNTech is set to seek approval for the use of its COVID 19 vaccine on children
-
Trials are currently being carried out to assert the safety of the vaccine of children between the age of 5 and 11
-
The company also disclosed plans to seek approval for the use of the vaccine in children aged 6 months to 2 years later this year
BioNTech is set to request approval for the use of its COVID 19 vaccine for children between the ages of 5 and 11.
The company is currently carrying out trials of the vaccine and results are expected soon.
Executives of the company told Del Spiegel that the results of their trial will be filed to health regulators to permits the use of the vaccine on children.
“Already over the next few weeks we will file the results of our trial in five to 11 year olds with regulators across the world and will request approval of the vaccine in this age group, also here in Europe,”
The latest statement gives BioNTech an edge over other vaccine producers in the race for children’s vaccine.
The company has revealed that a regulatory dossier will be filed on the kids between 5 and 11 as soon as September.
Read also: Another Deadly Disease Nipah Virus Threatens the World
In addition, the drug producer will go a step further to make the vaccine usable among kids between the ages of 6 months and 2 years by the end of the year.
Competitor Moderna had disclosed on Thursday that trial testing is being carried out for the use of its vaccine on children between 6 and 11.
Meanwhile, China has already begun using its own Sinovac vaccine on children as young as three in June.
Follow Naija.fm on our social media handles Facebook, Instagram, and Twitter to keep up with trending news, breaking news, entertainment news.





